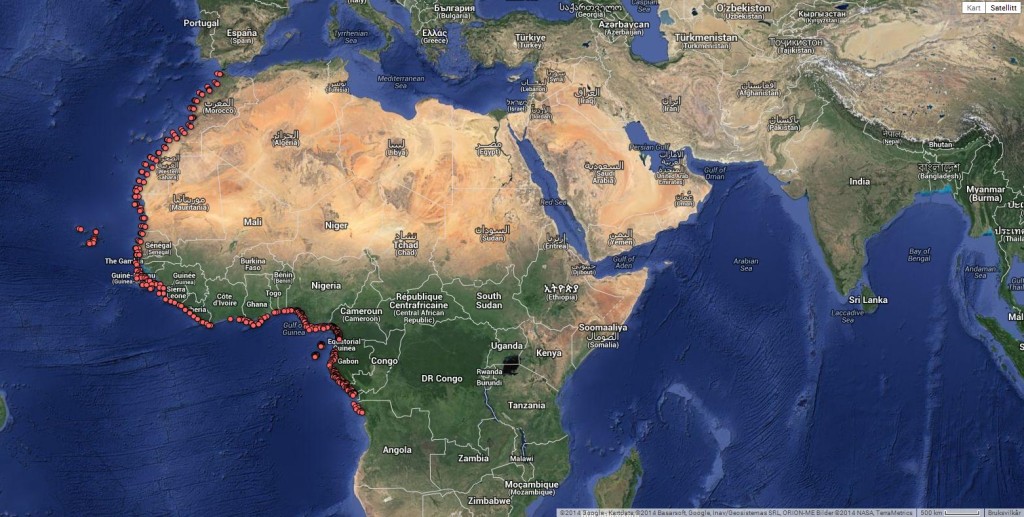
The study of the marine invertebrates of West Africa collected during the “Nansen Project” goes on and this year a second workshop was organized with a focus on the taxonomy of bivalves. Nine days of seclusion in our marine station at Espegrend on the surroundings of the city of Bergen allowed for the necessary tranquility to concentrate in the laborious work of sorting through and identifying thousands of specimens collected between Morocco and Angola by the Norwegian research vessel Dr. Fridtjof Nansen.
This year the “Molluscan” team was strengthen with two extra players; Sara Castillo, a PhD student from Spain enrolled at the University of Vigo and working on the marine fauna of Mauritania and Rudo von Cosel from the Paris Museum of Natural History a recognized authority on the taxonomy of bivalve molluscs.
During last year workshop we have implemented a successful “conveyor-belt operation” where each of us was responsible for a specific task (identification, labelling, databasing, imaging, barcoding, etc.). The samples were first organized by morphotypes and then passed into the hands and eyes of our taxonomic experts; a new label with a museum voucher number, species name, locality, etc., was added, and the samples were then databased, photographed, and some selected for DNA barcoding. In parallel several invited participants received training in the various technical and scientific aspects of this operation.
This year we have again implemented the same successful strategy; Lena Ohnheiser (University Museum of Bergen) was responsible for the database, labelling, and “in between” automontage imaging, Rudo von Cosel, José Pedro Borges (Portuguese Institute of Malacology), Kouakou Kouadio (University of Nangui Abrogoua, Côte d’Ivoire), Sidi Moctar (Institut Mauritanien de Recherches Océanographiques et des Pêches), and Sara Castillo for the taxonomic identification, and the author of these lines was the responsible for general imaging of the specimens and overall coordination of the team-work. In parallel Trond Oskars (a PhD student at the University Museum of Bergen) and Endre Willassen (PI of the Marine Invertebrates of Western Africa project) have worked on the preparation of 95 samples for DNA barcoding.
Our colleague Rudo von Cosel has been working for more than 10 years on a comprehensive book about the bivalves from tropical western Africa.
Rudo brought the proofs of his book to the workshop and we can proudly claim to have been the first ones to have ever seen his book assembled! The species illustrations and descriptions were bind together by families and used as identification tools during the workshop. At the end of the week we have databased over 800 lots and identified approximately 125 species. It was very rewarding to realize that our joint effort rendered many new geographical records contributing to better understand the distribution of species and biogeographic processes along the coast of West Africa.
We are very thankful to all participants in the workshop; to all those mentioned along these lines and those who were not but were nonetheless crucial for the success of this very productive week helping with various technical and logistic aspects, contributing to the good atmosphere, and very important keeping everybody “bellies” happy with great demonstrations of cuisine masterskills!
-Manuel